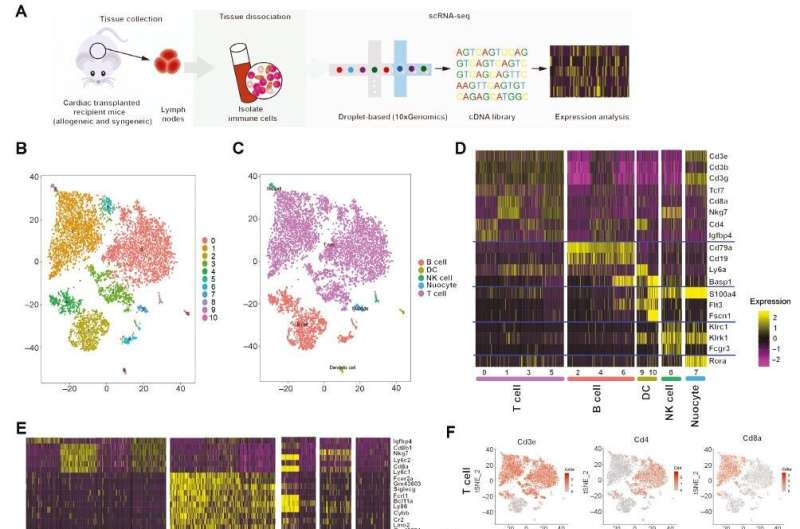
Immune cells play important roles in mediating allograft rejection and tolerance after cardiac transplantation. However, immune cell heterogeneity at the single-cell level, and how immune cell states shape transplantation immunity, remain incompletely characterized.
The authors of a new study, published in Cardiovascular Innovations and Applications, performed single-cell RNA sequencing (scRNA-seq) on immune cells in LNs from a mouse syngeneic and allogeneic cardiac transplantation model. Nine T cell clusters were identified through unsupervised analysis. Pathway enrichment analysis was used to explore the functional differences among cell subpopulations and to characterize the metabolic heterogeneity of T cells.
The transcriptional landscape of immune cells was determined, particularly T cells, and their metabolic transcriptomes in LNs during mouse cardiac transplantation. On the basis of molecular and functional properties, we also identified T cell types associated with transplantation-associated immune processes, including cytotoxic CD8+ T cells, activated conventional CD4+ T cells, and dysfunctional Tregs. The contribution of JunB to the induction of Th17 cell differentiation and restriction of Treg development was further elucidated, and the authors identified that HIF-1a participates in T cell metabolism and function.
The first systematic single-cell analysis of transcriptional variation within the T cell population is presented in this article, providing new insights for the development of novel therapeutic targets for allograft rejection.
More information:
Zhonghua Tong et al, Single-Cell RNA Sequencing Maps Immune Cell Heterogeneity in Mice with Allogeneic Cardiac Transplantation, Cardiovascular Innovations and Applications (2023). DOI: 10.15212/CVIA.2023.0023
Provided by
Compuscript Ltd
Citation:
Single-cell RNA sequencing maps immune cell heterogeneity in mice with allogeneic cardiac transplantation (2023, May 25)
retrieved 26 May 2023
from https://medicalxpress.com/news/2023-05-single-cell-rna-sequencing-immune-cell.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.




